Mechanisms underlying cell identity and plasticity
Using a combination of mouse genetic lineage tracing, fluorescence imaging, and single-cell RNA sequencing, we are investigating functionally distinct subsets of fibroblasts. These subsets exhibit unique capabilities to proliferate, differentiate, and secrete growth factors that support the growth of epithelial and endothelial cells. Our goal is to identify the signaling pathways and transcription factors that regulate fibroblast differentiation.
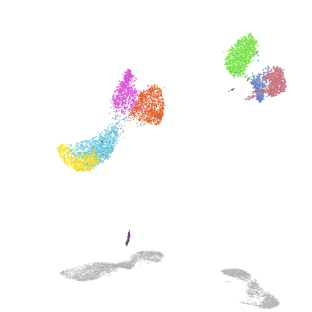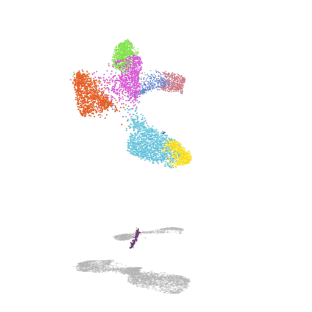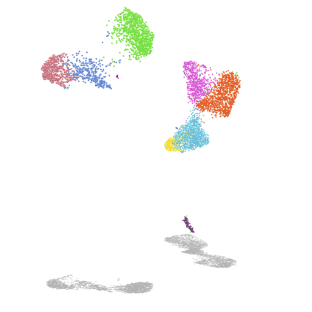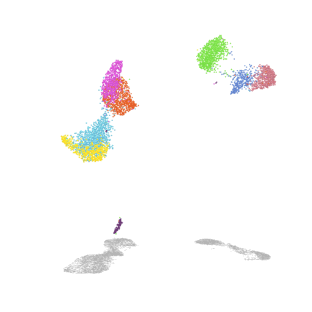
Mechanisms of alveolar development
Our lab uses mouse genetics and single-cell RNA/ATAC sequencing to investigate how mesenchymal lineages contribute to lung development. We are also developing new assays to examine the diverse functions of mesenchymal cell subsets, including assays for cell migration and force exertion. We are also exploring how mesenchymal and immune lineages co-develop during the early stages of environmental exposure.
Translational impact for lung physiology and disease
Our approach is to uncover the mechanisms that regulate unique cellular processes and to determine how these processes influence lung function, disease onset, and progression. To assess outcomes in our murine genetic models, we use physiological parameters and in vivo systems that elicit specific cell behaviors under conditions of tissue stress. In addition, we collaborate with researchers across the CHOP and Penn communities to obtain clinical samples and primary cells for validating novel markers and cell phenotypes.