March 2021 – New paper . . .
“Genomic, epigenomic, and biophysical cues controlling the emergence of the lung alveolus”
Journal Link: Science
LungMAP Explorer: Mm Timecourse
Raw Sequencing Data: Geo DataSet
GitHub Links: Seurat, Morrisey Lab
In our recent study we used single-cell RNA sequencing (scRNA-seq) and in vivo mouse genetics to identify factors expressed between cell-types that facilitate cellular maturation and lung alveolar growth.
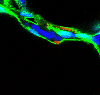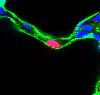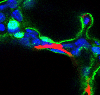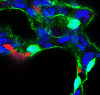
The mouse scRNA-seq atlas showed that alveolar type 1 (AT1) epithelial cell lineage had enriched expression of several ligand (secreted factor) encoded RNA transcripts (Figure 1). Further, we identified the cognate receptors for these ligands in a subset of fibroblast, termed the secondary crest myofibroblast (SCMF). Using 3D imaging of the developing lung, we show that AT1 cells are in close proximity to the SCMF-precursor during embryonic development, indicating that these cells are aligned in a spatial and temporal manner (Figure 2).


The alveolar region of the lung is where gas-exchange, the transfer of CO2 and O2, occurs. Individual alveoli are lined by the elongated AT1 cell (Figure 3). To form this delicate tissue the lung goes through a developmental process referred to as alveologenesis (the generation of alveoli). This process is the final step in lung maturation and spans the period around birth and into early adolescence.
To shape and expand the overall surface area of the alveoli which will enhance gas-exchange efficiency, the myofibroblast or SCMF, exists transiently during this stage and is thought to contribute to sculpting the nascent alveolar tissue (Figure 3). We demonstrate that the alveoli has two sub-types of Pdgfra-expressing fibroblasts, the SCMF and a Wnt2-expressing fibroblast. Using genetic reporters we purified these two fibroblasts and show that the SCMF is unique in its ability to exert force. Lastly, we use AT1 cell-specific conditional knock-out mice to examine the effects of AT1-derived ligands, Wnts (Wnt7a/b, Wnt3) and sonic hedgehog (Shh), in regulating the differentiation of the SCMF in the post-natal lung. Loss of Shh from the developing AT1- cell (Hopx-expressing) leads to impaired formation of SCMF cells and dilated alveoli.